If your reading this post you probably already have an understanding of a few basic concepts. For those who missed it here they are for review.
Basic Concepts
- A dyne per centimeter is a unit of force
- A dyne is defined as the force required to accelerate a mass of one gram at a rate of one centimeter per second squared
- A dyne per centimeter is the unit traditionally used to measure surface tension and interfacial tension
- Surface tension is a measurement taken on a liquid and Interfacial tension is a measurement taken between/among liquids.
- Surface tension and Interfacial tension are both measured using an instrument called a tensiometer
I suppose it's worth mentioning why we decided to write this particular post about tensiometers and their surface tension and interfacial tension measuring limitations. The simple explanation being that people have continued to ask us the question! In fact, one time we had someone ask if we could recommend a tensiometer that could measure up to and above 500 dynes per centimeter! ...and we simply said "why?" and scratched our heads.
I guess you could say we finally received enough inquiries about this to force us into action and write about it.
Tensiometer readout design
If you have ever looked at a manual tensiometer guage like this one you will notice the dial only goes to 90 (90 dynes per centimeter).

Compare this to some of the automatic tensiometers that use sensitive electronic balances and software and you will notice they have for the most part (various models) a range up to around 100 (100 dynes per centimeter). See the red arrows below;
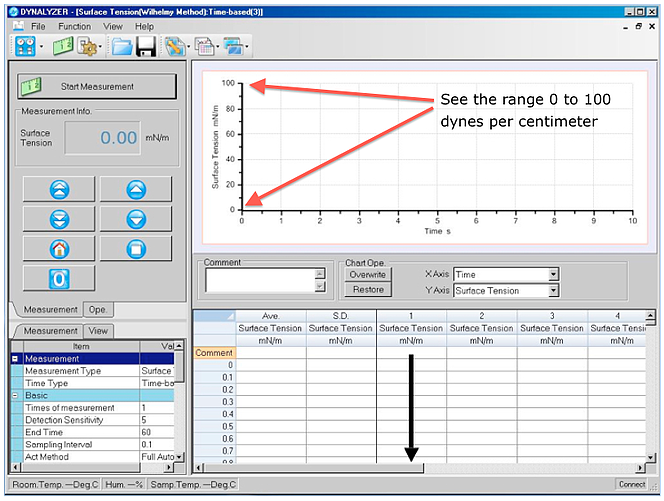
Why not make them with larger ranges?
Up to this point you have seen a couple of examples of the range capabilities for a manual and an automatic tensiometer. The truth of the matter is that the dial on the manual tensiometer could be re-etched to include additional numbers going up much higher than 90. And the automatic tensiometers using the electronic balances could be configured and the software re-written to go as high as 1,000!
So why haven't the engineers who make these tensiometers made the range on their tensiometers as big as they can? Wouldn't that make the tensiometer that much more appealing? A bigger range would mean you could measure more samples right?
Well maybe...but not on this planet or in this universe.
Liquids and fluidic metals?
You see, the answer is not really about the tensiometer. In general, except for fluidic metals there are no known liquids that will indicate a surface or interfacial tension above 90/100 dynes per centimeter. Surface tension of Mercury for example is generally reported around 480 dynes per centimeter but it does not wet to either the Wilhelmy Plate or Du Nouy Ring and cannot be measured by a traditional tensiometer. Other fluidic metals need high temperatures and special atmoshperic conditions and are also unsuitable for traditional tensiometers. With the exception of fluidic metals just mentioned solutions including metal ions but excluding surfactants (e.g. plating solutions) indicate comparatively high surface tension readings of 80 to 90 dynes per centimeter at most. One of the highest surface tension liquids except Mercury is Sodium Chloride 6.0mol/20°C at 82.55 dynes per centimeter (mN/m).
In summary then, we can conclude that all of the liquids we will encounter while measuring surface tension or interfacial tension with a traditional tenstiometer will fall in the range not to exceed 100 dynes per centimeter. Therefore restating the obvious you don't need a tensiometer that can measure surface tension above 100 dynes per centimeter.
Make sense?











 We all know that surface tension affects our daily lives thru everyday applications like the ink you use in a pen, detergents for washing clothes, soap to clean your hands, paint for the house, just to name a few. But Surface tension is more involved in your life than you may think.
We all know that surface tension affects our daily lives thru everyday applications like the ink you use in a pen, detergents for washing clothes, soap to clean your hands, paint for the house, just to name a few. But Surface tension is more involved in your life than you may think.